 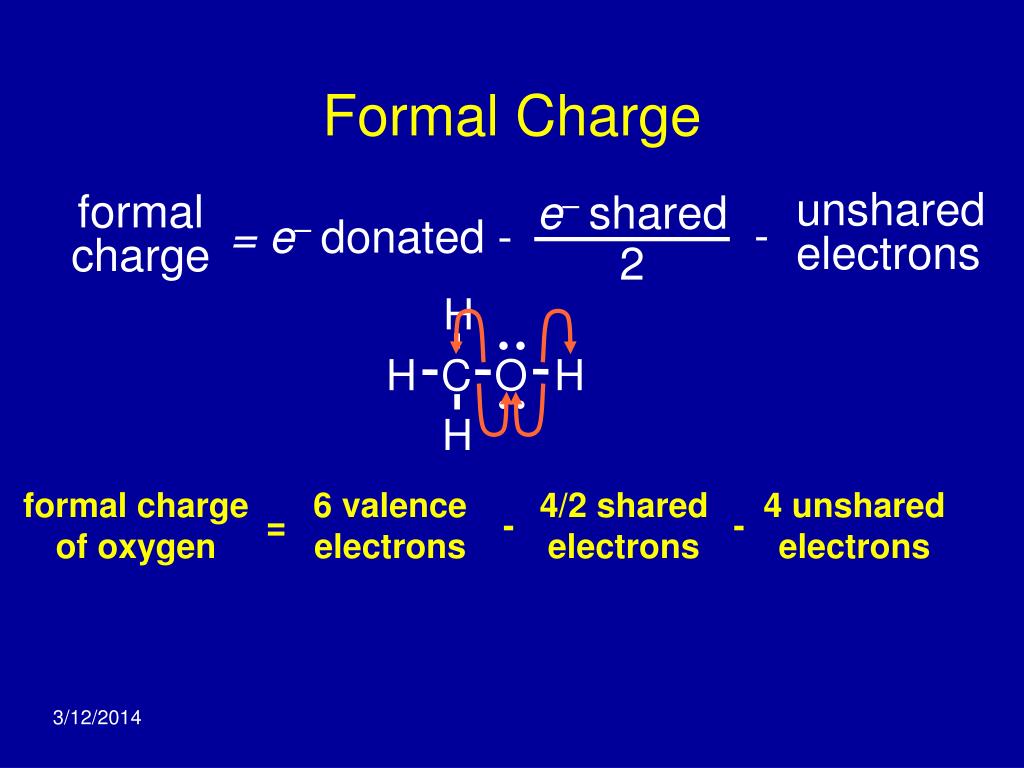
Conventionally the net charge is written with the magnitude before the sign the magnitude of singly charged molecules/atoms is generally omitted.They are repelled by like electric charges and are attracted to opposite charges.The positive electric charge of a proton is equal in magnitude to the negative charge of an electron therefore, the net electric charge of an ion is equal to its number of protons minus its number of electrons.Discuss the relationship between charge transfer and the color of a metal complex. 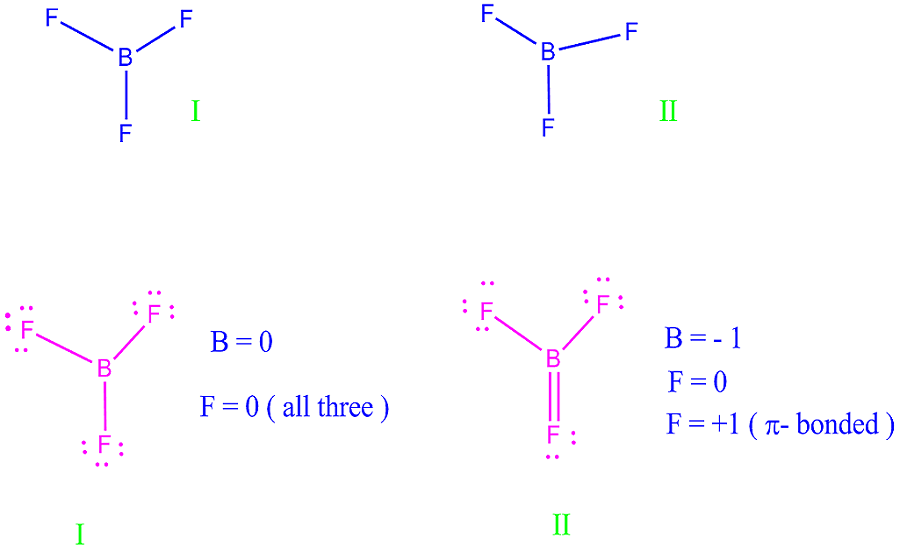
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
